Our Team

Who We Are
“Nippon Shokubai has a long-term approach. That is why we stand behind you as you help build the business.”
These words were how a board member described the company to me in 2019, seven years after Nippon Shokubai had entered the life science sector.
Already, the life sciences have changed incredibly. Super-aging societies are accelerating, putting pressure on the development of new modalities such as regenerative medicine. It is in this environment that the Health & Medical Business Division of Nippon Shokubai built an oligonucleotide and peptide API synthesis facility in 2019. Since then, we have accelerated the establishment of our CDMO systems, and in 2021, we made our first shipment of GMP APIs.
In just 10 years, Nippon Shokubai, a leading synthetic chemical company, has established a mass production system of oligonucleotide and peptide APIs. There are two reasons for this success.
One is the attitude of nurturing the business patiently, as stated above. The other reason is the “insatiable curiosity, inquiring minds” all our employees have. We entered this sector with limited knowledge or experience in drug discovery. However, we had confidence in our employees. I was surprised how quickly our business grew into a CDMO, built elaborate research facilities, and initiated active partnerships with academia and venture development companies.
Underlying our rapid growth is our mission of “TechnoAmenity: Providing prosperity and comfort to people and society, with our unique technology”. It is our corporate culture to develop Amenity through the power of Techno ー constantly using evolving technology to benefit people and society.
The life sciences are testing human ingenuity for a better world. Nippon Shokubai is contributing to society with this goal in mind. As a team that “creates value through technology,” we would be happy to work together as partners.

Shingo Horimoto, Ph.D.
Director of Health & Medical Business Division

Our Goal
Nippon Shokubai supports human health and medical care and contributes to the future of society by supplying oligonucleotide and peptide APIs.
Oligonucleotide and peptide APIs are a new modality that fill a gap caused by the limitations of low molecule and high molecule (antibody) drugs.
We provide solutions in oligonucleotide and peptide medicines and DDS markets by having established a leading GMP system in Japan, unique technologies, and a one-stop shop service system from exploratory research to clinical trials, thereby contributing to therapeutic technologies in disease areas where treatment satisfaction is low.


Approach
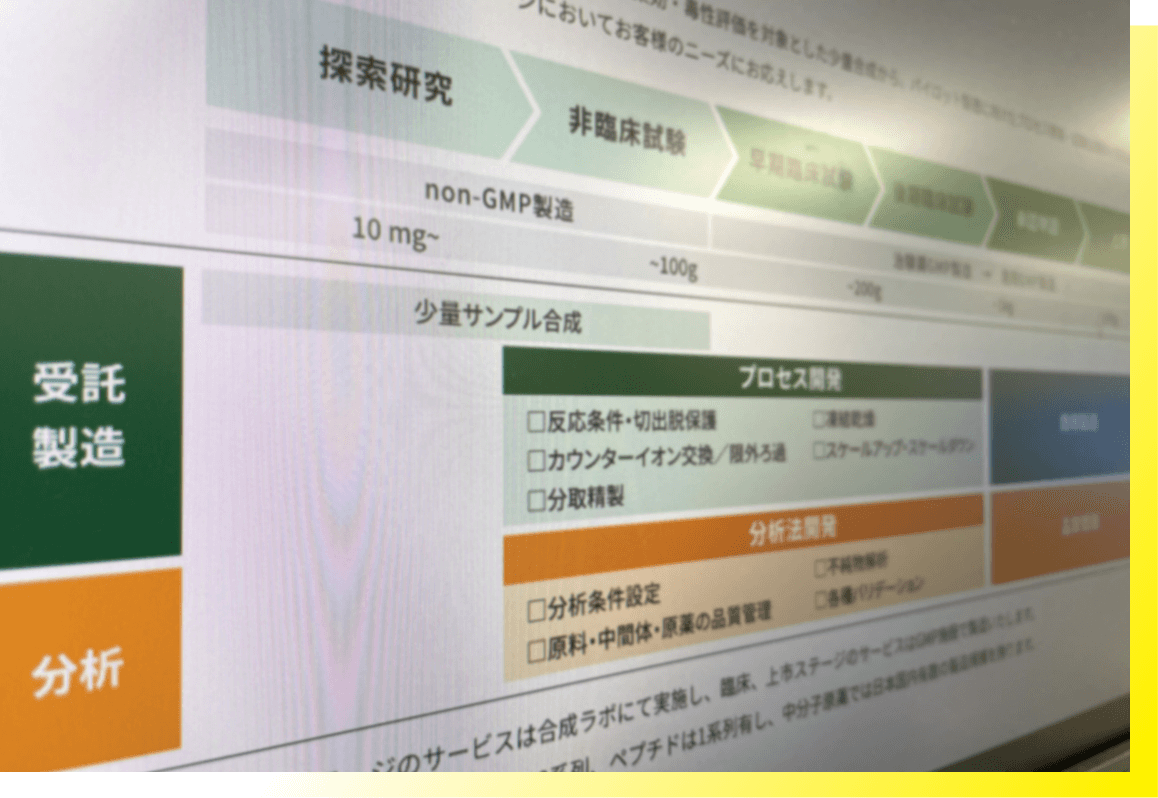
Providing a wide range of manufacturing
We have established a system suitable for exploratory research to clinical trials, which enables us to offer solutions ranging from small-scale synthesis for drug efficacy and toxicity evaluation in the drug discovery stage, to contract research on processes and testing methods for pilot manufacturing, and GMP-compliant API manufacturing for clinical trials.
Also, we offer a variety of manufacturing scales ranging from a few µg in the exploratory research stage to 500 g in the late clinical stage.

Active collaborations with promising venture companies and academia
Since forming a capital and business alliance with GlyTech, Inc. in 2015 to jointly advance manufacturing process development and clinical trials, we have actively expanded collaborations with leading domestic biotech ventures. In 2016, we entered into a capital alliance with TAK-Circulator Corporation, followed by the acquisition of Rena Therapeutics Inc. in 2019 and Lilac pharma Inc. in 2024. Through these and other diverse initiatives, we strive to advance pioneering research and the development of unique technologies.

Members
Our experienced professionals work as a team for the success of your project.
R&D (synthesis, analysis, DDS)
-

Jun Kotera
General Manager
-

Masayuki Utsugi
Group Leader
GMP Manufacturing
-

Masahiro Shiroshima
Group Leader
-

Hiroaki Terashi
Manufacturing Supervisor
-

Kiyoshi Nose
Manufacturing Unit Manager
-

Yoshimasa Yanamoto
Quality Control Unit Manager
-

Yoshiki Mori
Quality Assurance Unit Manager
-

Hiromichi Tanaka
Facilities Maintenance Unit Manager

Our History

| August 2012 | Established Health & Medical Business Preparation Office |
|---|---|
| October 2015 | Started joint process development & joint clinical development with GlyTech |
| October 2016 | Built the Synthesis Laboratory at the Suita Research Center |
| January 2019 | Built a GMP facility |
| September 2019 | Started the joint commercialization project of TAKC-02 (development code) with TAK-Circulator |
| November 2019 | Made Rena Therapeutics a subsidiary |
| February 2020 | Established the Development Promotion Center (Shonan) |
| March 2021 | Shipped our first GMP lot of API |
| April 2021 | Started full-scale sales operations |
| April 2025 | The Development Promotion Center was closed and Tokyo Sales Office was newly established |

Locations
-

Laboratory (Osaka)
-

GMP facility (Osaka)
-

Osaka Sales Office
-

Tokyo Sales Office



